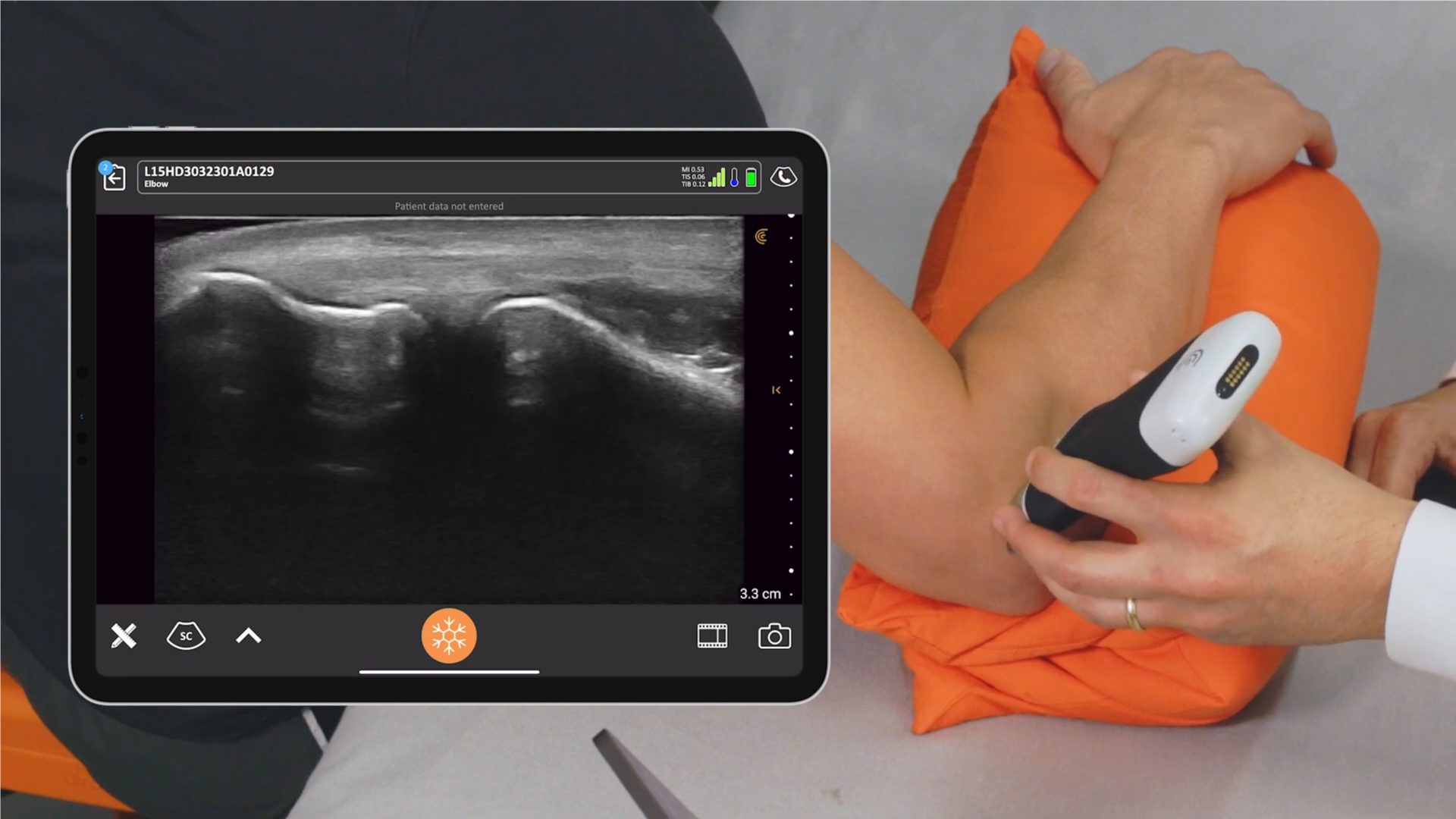
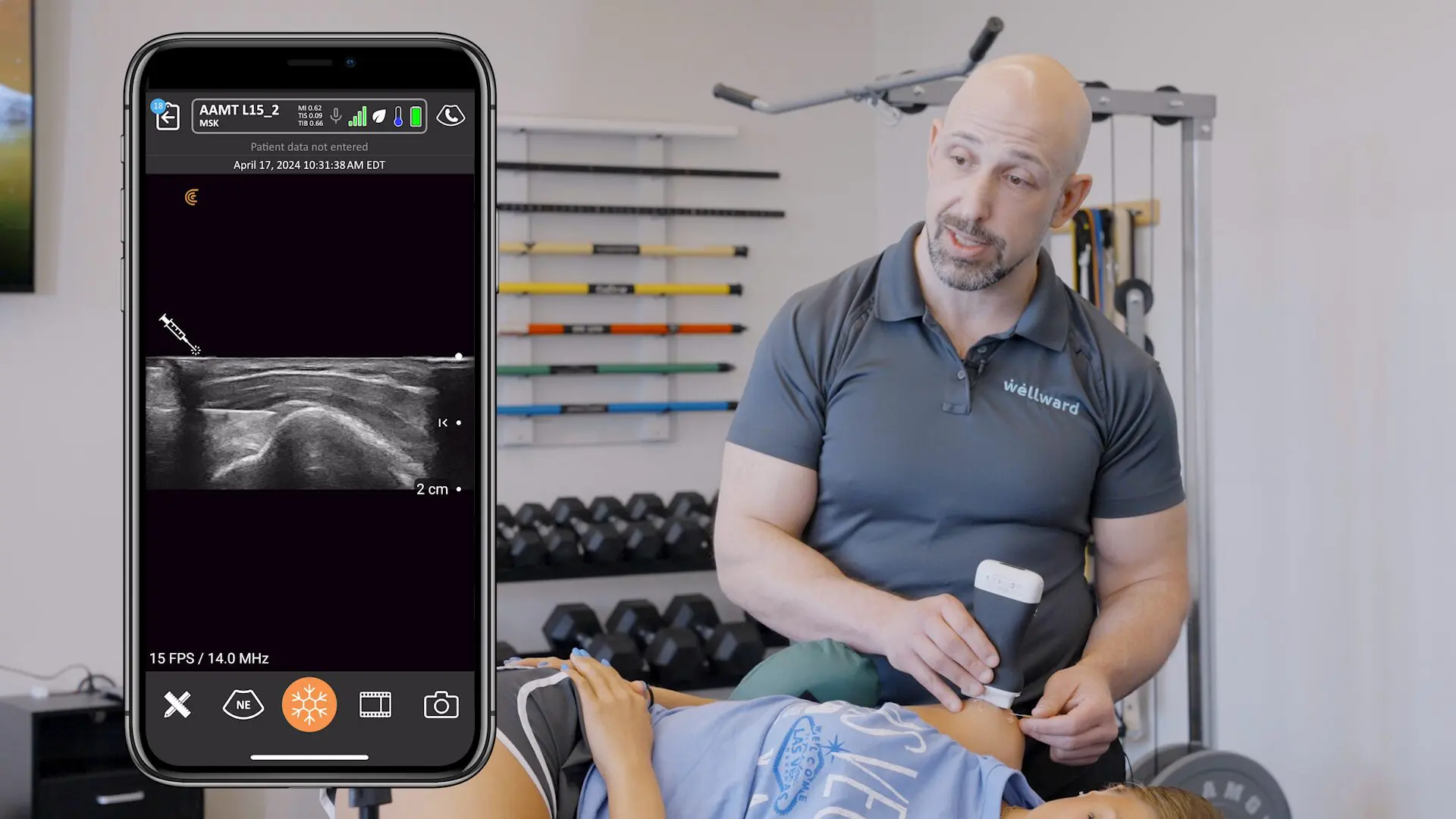
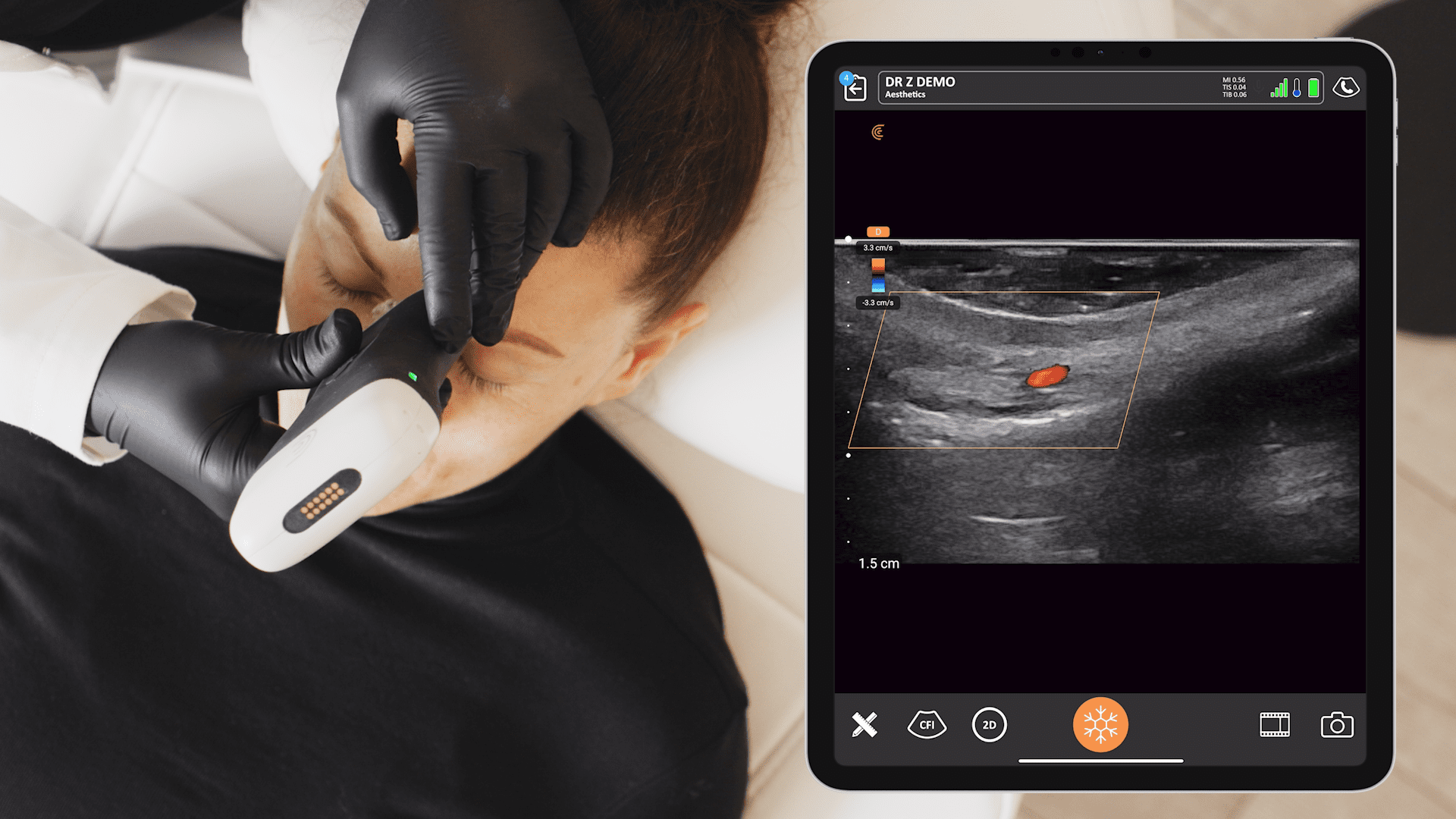
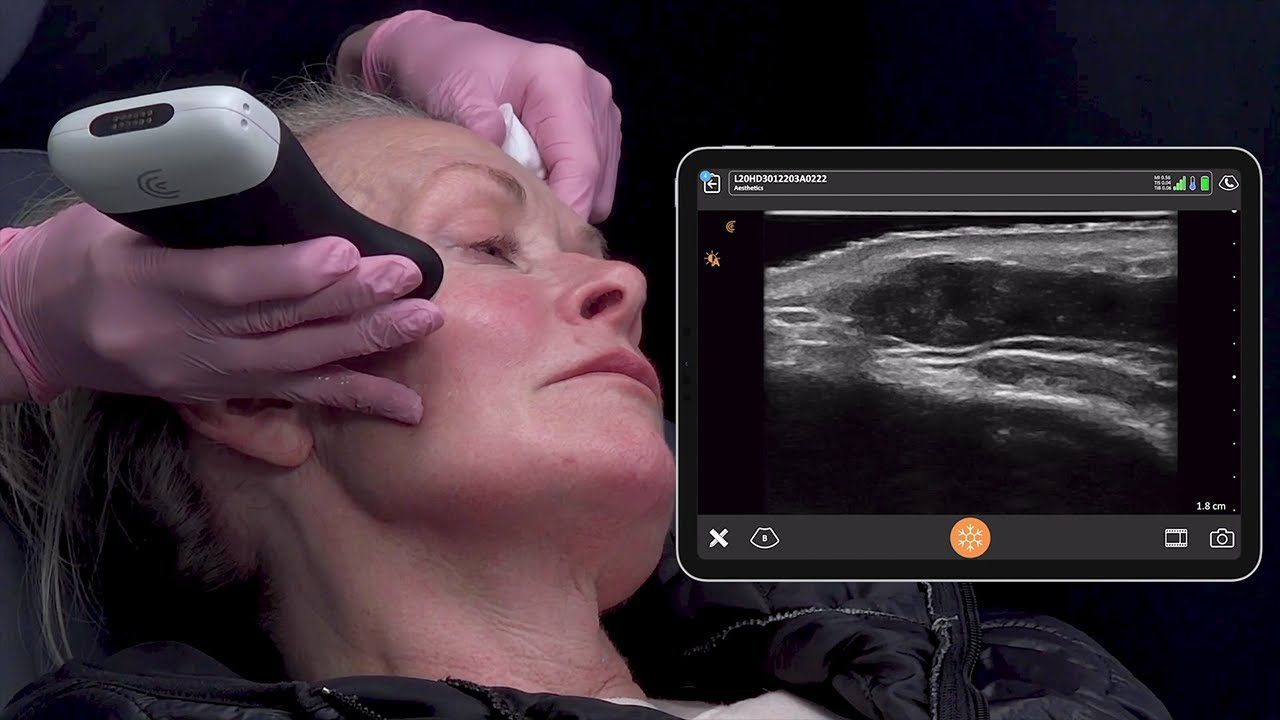
Last Saturday, key members of the Clarius management team woke up to find an exclamation-mark-filled e-mail message in their inboxes. The message came from Abhijit Ahir, Clarius’ head of Regulatory and Quality Affairs.
Coming from the usually reserved Abhijit, the tone of the e-mail clearly indicated his surprise. Fortunately, it was the good kind of unexpected news.
Abhijit had just learned that Clarius had obtained its ISO 13485 certification a few weeks earlier than expected. ISO 13485 is a globally-accepted mark of quality for medical device manufacturers and brings Clarius another step closer to regulatory approval.
“Getting ISO-certified might not sound that exciting to a layperson, but it’s actually a really important milestone for Clarius,” said Abhijit.
Abhijit has eight years of experience in quality assurance and has worked on many different quality standards, including ISO, CMMI and COBIT. He explained that the ISO 13485 certification is especially important for medical device companies.
“Think of it as a ticket to the world,” he said. “ISO 13485 is required in many jurisdictions, such as Canada and the European Union, so the certification is like an international stamp of approval.”
ISO 13485 is a requirement for obtaining regulatory clearance in many countries. It validates the rigor of a company’s quality management systems, proving that it is capable of producing a consistent, high-quality product.
It is not a distinction that comes easily. Abhijit’s team had to thoroughly review all of Clarius’ internal procedures, ensuring that they measured up to ISO’s exacting standards. The process culminated in an on-site inspection by ISO, where every single policy and practice was scrutinized in painstaking detail.
“The goal is always to think of everything beforehand, so you can give the auditors the answer they need without them having to ask,” remarked Abhijit. “That’s sometimes easier said than done, but in this case, the team did a great job.”
With the ISO certification hurdle crossed, the quality team is now focused on obtaining regulatory approval in jurisdictions like the U.S., Canada, the E.U. and China.
Abhijit is proud of what the team has accomplished in a short period, and is already looking towards the next step.
“Actually, this is not just a validation of the company, but also of the quality team’s abilities,” he said. “The discipline we’ve developed here leaves us well-equipped to work toward future regulatory milestones.”