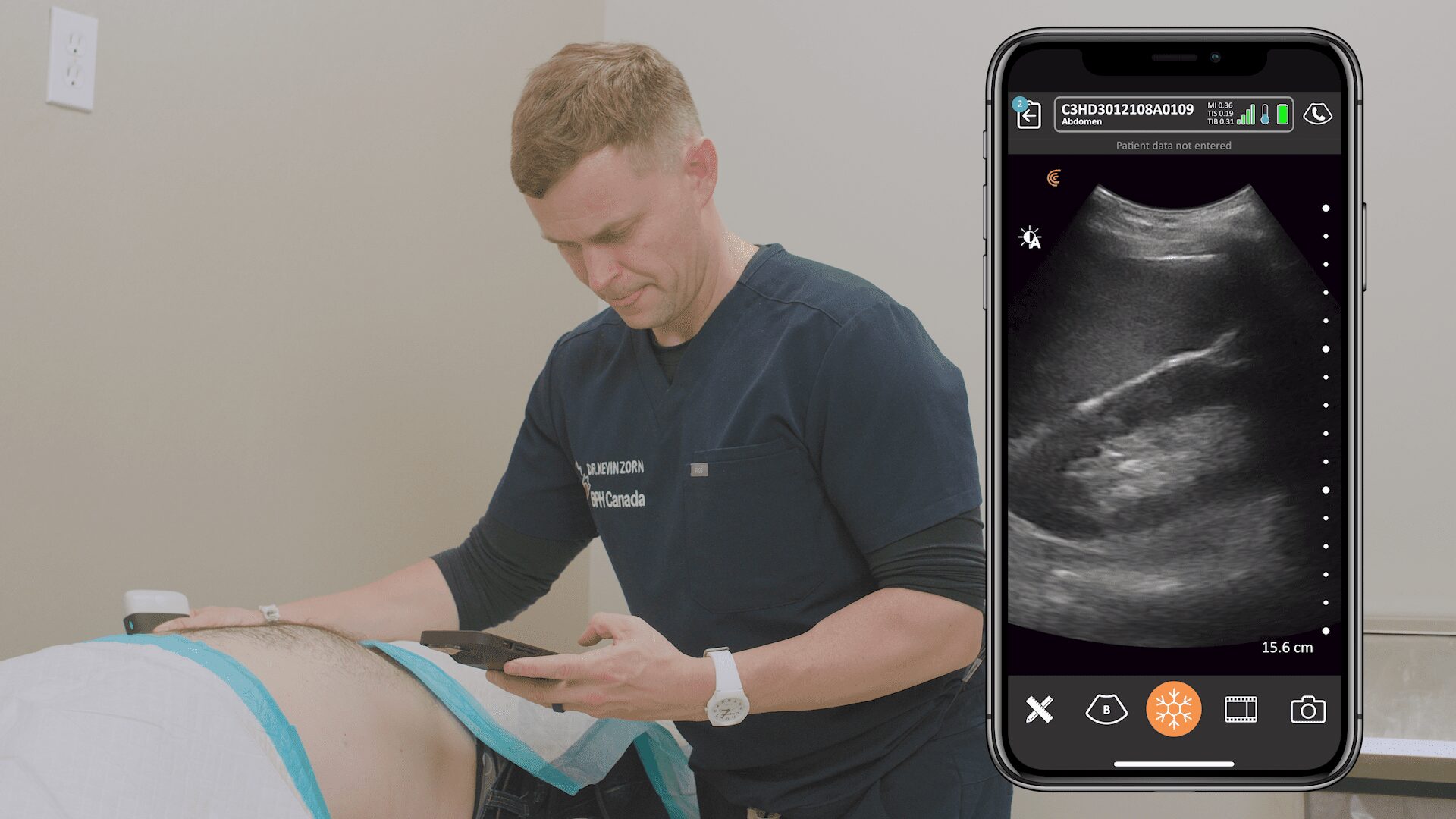
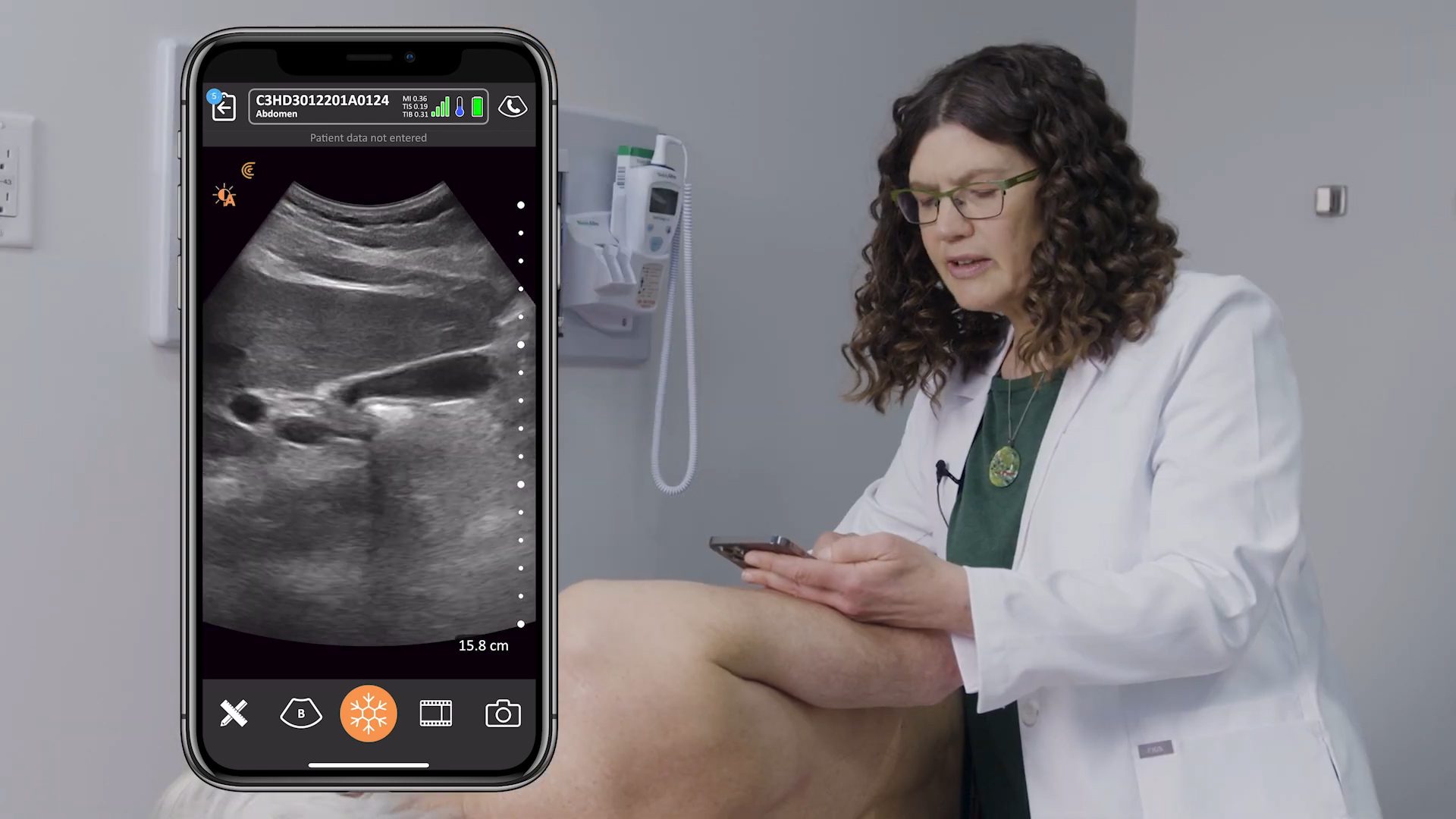
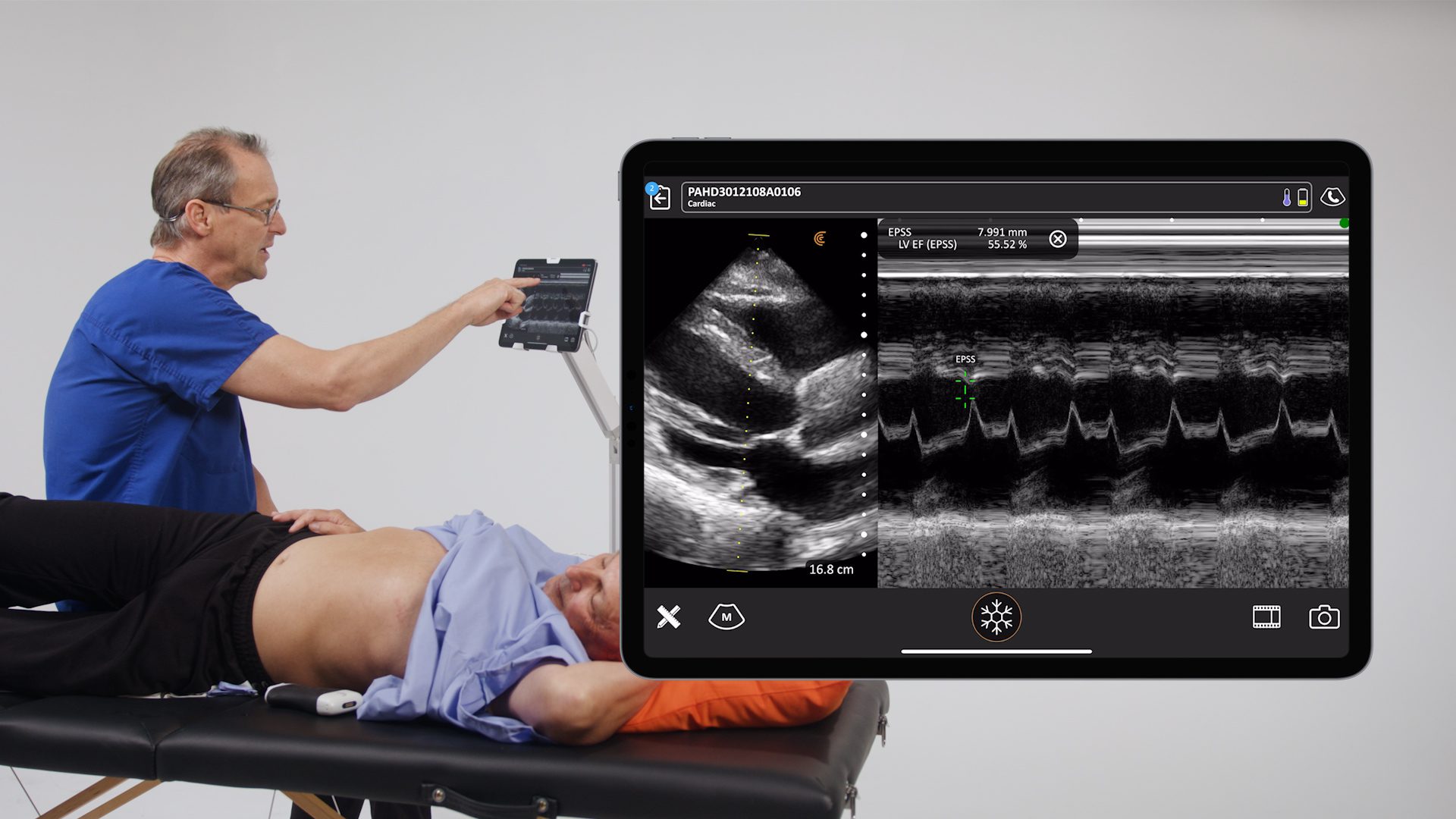
We’re delighted to receive 510(k) clearance from the U.S. Food and Drug Administration (FDA) for our new Clarius Bladder AI, a non-invasive tool that automatically measures bladder volume in seconds. It’s available now in the United States with the Clarius PAL HD3, Clarius PA HD3, and the Clarius C3 HD3 wireless handheld ultrasound scanners.
Clarius Bladder AI is included among other advanced software features that are available with the Advanced Primary / Critical Care Package, included with Clarius Membership. Current Clarius members with eligible scanners will be able to use the new application on their Clarius scanners today.
Bladder AI removes the tedious steps of calculating bladder volume in my patients with urinary retention,” says Dr. Oron Frenkel, an emergency physician who practices in the United States and Canada. “It helps me quickly identify who needs a catheter placed or further investigation for any urinary or abdominal pain symptoms.”
Watch this video to see Bladder AI in action:
According to a 2023 published study, “bladder ultrasound in clinical practice is feasible and reduced the inappropriate use of catheters, reducing patient risks and healthcare costs.”
The latest addition to the Clarius AI portfolio, Clarius Bladder AI accurately measures bladder volume and provides real-time feedback to clinicians. It is particularly useful for monitoring urinary retention and assessing bladder emptying in patients with neurogenic bladder or urinary tract obstruction.
In contrast to dedicated bladder scanners priced at approximately 15,000 USD, Clarius offers an affordable all-in-one solution that integrates bladder scanning with whole-body HD ultrasound for abdominal, pelvic, and thoracic imaging, and for guiding precise IV access. Healthcare institutions can save on bladder scanning equipment by purchasing Clarius scanners starting at 3,595 USD plus annual membership.
We made the decision to develop Clarius Bladder AI in response to the increasing demand from health systems for multi-function devices that can be used by physicians, nurses, and other ancillary staff for ultrasound-guided peripheral IV insertions and bladder scanning,” says Kris Dickie, Clarius Chief Technology Officer. “Paired with our new Clarius PAL HD3, which offers the capability to image deep anatomy and superficial anatomy, it’s a more cost-effective and versatile tool than current bladder scanners.”
Clinical Applications for Clarius Bladder AI
Acute Care – Ultrasound is used to measure postvoid residual urine, and evaluate for bladder and urethral stones, hydronephrosis, and upper urinary tract disease. Bladder ultrasonography is non-invasive and comfortable for the patient and reduces the need for catheterization.
Urology – Urology clinics in the United States across the country receive around 21 million patient visits, primarily due to bladder retention, a condition associated with various complications. Bladder ultrasound plays a crucial role in enhancing diagnostic accuracy by assessing bladder volume and contributing to an accurate initial diagnosis during the first visit.
Nursing Homes – “Literature suggests that more than 70% of nursing home residents in the United States experience UI (urinary incontinence)”. In recent years, bladder ultrasonography has become widely used for successful evaluation and management of UI, rehabilitation, and a complete assessment of bladder function in outpatient settings without the need for invasive catheterization.
Book a Demo of Bladder AI
Interested in using Clarius ultrasound with Bladder AI for your practice? Book a private virtual demonstration with a Clarius expert today. If you’re already a Clarius user without membership, we would be happy to discuss the benefits of adding membership for a small annual fee, which includes access to Bladder AI, Cloud Storage, and much more.